What is Radiation?
To many, the term radiation is a confusing and even a terrifying subject, but it need not be so. Like any other subject, once its better understood, it’s not as mysterious or frightening.
The purpose of this article is to present this subject as simply as possible by avoiding nuclear physics, mathematical formulas, and complex scientific explanations. I’ve also added some great YouTube video links that further support the basic concepts presented.
While the term radiation is commonly associated with radioactivity, in the purest sense, it can be any type of energy radiating from its source.
Different kinds of radiation would include thermal radiation from the sun, acoustical radiation such as ultrasound or music from a radio speaker, and nuclear radiation resulting from naturally radioactive materials or man-made processes, etc.
For the purpose of this article I will focus on the nuclear type of energy, and more specifically, the kind that can cause bodily harm. Let’s begin with a high-level definition as follows:
Radiation is defined as the emission of energy in the form of electromagnetic waves or subatomic particles that cause ionization.
Wikipedia
So let’s break this definition down into its separate 4 parts so we can attempt to better understand what this really means:
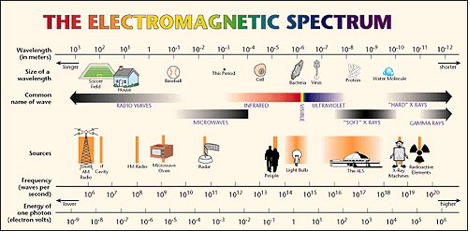
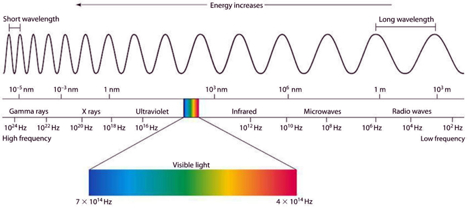

Background Radiation
This does not mean that all ionizing radiation is bad or going to kill us. We are surrounded by ionizing radiation rising from the earth’s geological formations, some of the foods we eat, and from the cosmos above. Even our bodies contain some elements that are radioactive, albeit very miniscule amounts.
Background radiation is what we hear whenever we hear the clicking sounds coming from a Geiger counter even when no extraneous or known radiation source is present. The background ionizing radiation varies from one location to another depending upon the geological formation. Granite for instance, emanates a fair amount of natural radiation that results in higher background radiation readings.
Likewise, cosmic radiation streaming from the heavens above are continually filtered by the earth’s atmosphere until it reaches the surface. Those living at sea level typically have the lowest background levels when compared to those living at higher altitudes like Denver. When traveling at 35,000 feet in an airliner, passengers will experience radiation levels 40 to 60 times higher than when at sea level. Astronauts traveling in space are the most vulnerable since they are not being shielded by the earth’s atmosphere.
Another natural radiation source is radon which is released from the ground into the atmosphere. This inert type of radioactive gas is odorless, colorless, and tasteless so like other radiation types it cannot be detected by our human senses. When radon becomes trapped inside home basements the concentration levels can rise to dangerous levels. Studies have linked breathing high concentrations of radon to lung cancer.

metaphor for how radiation behaves
Radiation Behavior
The sun is a great teacher to better help us understand how Radiation behaves. If we traveled inside a spaceship and got too close to the sun we would certainly die. Yet, back on earth simple shielding can soften or even eliminate its adverse effects. If we limit the time we are exposed to the sun’s brilliant rays, we can avoid sunburns. And sometime in the distant future, the sun will eventually burn up all its fuel and will no longer radiate.
Radioactive materials we typically see in industrial applications all follow these same three principles of distance, shielding, and time. When radioactive materials like Cs-137 and Co-60 are properly shielded and kept at a safe distance, their purposeful design ensures we remain safe. Even when we are in close proximity while performing maintenance, by limiting our time, we can still remain safe. Distance, shielding, and time are the three corner stones to any radiation safety program and when applied properly will ensure everyone’s dose is well below regulatory limits.
Another well-known behavior is that radioactive materials give up all their energy over time through a decay process until they become stabilized and are no longer radioactive. The time to stabilization or equilibrium varies by element and ranges from fractions of a second to billions of years. A measurement of time used in the nuclear field to clock the decay process is called the half-life, or the time it takes to decay half of its activity. Cs-137 has a half-life of 30 years, so after 30 years, the radiation being emitted is only half of what it was when measured at the time it was installed in the device. After about 7 half-lives, the material reaches its equilibrium state. Thanks to modern-day microprocessors and memory, modern radiation devices can compute the decayed value on a daily basis and re-calibrate itself to compensate for the ever-decreasing output of the radioactive source. Never-the-less, at some point well before its total demise, the radiation output becomes too weak to perform its primary function and must be replaced either by a new source or device.

Beneficial Uses of Radiation Go Largely Ignored
Mankind has found many truly beneficial uses for radiation in enhancing our lives. Unfortunately, radiation gets a lot of bad press resulting from the use of atom bombs in WWII, the nuclear reactor disasters at Chernobyl and more recently at Fukushima, and the prospects of dirty bombs by terrorists. Early adoption of radioactive products and elixirs shortly after radioactivity was discovered and before its harmful effects were known also did not help its cause. We hardly ever hear about positive uses of radiation outside of nuclear medicine and nuclear power; and even those are controversial in their own ways.
Thanks to the properties of radiation, mankind has found numerous other beneficial uses that include:

Nuclear Gauges
Nuclear gauges that measure materials being processed for density, moisture, flow rate, and material levels in a wide variety of industries including paper & pulp, chemical/petrochemical, steel, food, packaging, roofing products, mining, fertilizer, construction and many more.

Products
Products including smoke detectors, x-ray machines used in non-medical applications like those built to perform quality inspections on welds, CT scanners, exit signs, static eliminators, depleted uranium penetrators

Irradiators
Irradiators that sterilize fruits, vegetables, and other food products, medical and pharmaceutical supplies, eradicating insects through sterile male release programs, calibrating dosimeters and radiation detection instruments, security applications

Research
Research for discovering material age via carbon dating, tracking living organisms by injecting radioactive tracer materials, studying environmental pathways in a similar manner, changing material composition
Most of us are touched in one way or another on almost a daily basis by some process or product that employs radiation. Despite what many might think, the uses of radiation are expanding as new technologies and materials continue to evolve.
Conclusion

We need not look beyond our own sun to grasp the basic principles of radiation. While ionizing radiation is fundamentally simple to grasp, it becomes immensely complex and is still being studied at research institutes world-over. Pin-pointing the precise radiation level at which radiation is beneficial and where it becomes harmful has been a point of great debate for many decades with no clear ending in sight.
The discovery of radiation at the end of the 19th century opened our eyes to the understanding of elements here on earth and enables us to detect distant galaxies far beyond our natural eye-sight. It is amazing, powerful, dangerous, and beautiful all at the same time. Harnessing this great power has changed our world forever and will undoubtedly continue to open new vistas far into the future.
